 Dierssen Lab
Dierssen Lab
 Systems and Synthetic Biology
Systems and Synthetic Biology
- Group page
- Research lines
- Group members
- Publications
- Neuroscience Resources
1989 Ph.D. Department of Physiology and Pharmacology, Universidad de Cantabria (Spain).
1989-1990 Research fellow, Freie Universität Berlin
1990-1993 Postdoctoral work at the Universidad Autònoma de Barcelona (Spain).
1993-1997 Assistant Professor at the Universidad de Cantabria (Spain).
1997-2001 Research position. Medical and Molecular Genetics Center-Institut de Recerca Oncológica (IRO), Barcelona (Spain).
1998-2007 Professor of Psychobiology at the University Ramon Llull
2001-Present Group Leader at the Center for Genomic Regulation, Barcelona (Spain).
Group news
Down syndrome linked to changes in mobile DNA fragments (25/07/2024)
A study demonstrates for the first time that retrotransposons, DNA fragments which can move around the genome and alter gene expression, are abnormally expressed in Down syndrome.
Learning and Memory Problems in Down syndrome Linked to Alterations in Genome's ‘Dark Matter’ (27/02/2024)
Researchers at the Centre for Genomic Regulation (CRG) reveal that the Snhg11 gene is critical for the function and formation of neurons in the hippocampus. Experiments with mice and human tissues revealed the gene is less active in brains with Down syndrome, potentially contributing to the memory deficits observed in people living with the condition.
€2.7 million to begin Phase IIa trials for CTH120 in Fragile X syndrome (12/02/2024)
The public-private consortium made up of biotechnology company CONNECTA Therapeutics, the CRG, the IMIM and the I3PT, has been awarded €2.7 million in funding from the Spanish Ministry of Science, Innovation and Universities and the European Union through the NextGeneration EU programme to start Phase IIa clinical trials for CTH120 in the treatment of Fragile X syndrome (FXS).
Mara Dierssen, elected fellow of the Royal Academy of Medicine of Catalonia
Connecta Therapeutics starts clinical trials for its Fragile X syndrome drug (June 2023)
A Phase I clinical trial will assess the safety and tolerability of CTH120 in FXS. CTH120 showed very promising results in preclinical studies carried out at Dierssen lab
Antiretroviral drug improves cognition in a mouse model of Down syndrome (28/06/2022)
This study pioneered the discovery of lamivudine as a new therapy for intellectual disability and reveals retrotransposition as an interesting mechanism to pursue in neurodevelopmental disorders
International study finds increased mortality in adults with Down syndrome (22/02/2021)
Findings of the Trisomy 21 Research Society, led by Prof. Mara Dierssen, reiterate the need to prioritise vaccinations for Down syndrome
People with Down syndrome are genetically susceptible to COVID-19 (09/02/2021)
A study by Dierssen's lab reveals the genetic factors involved in the predisposition of people with Down syndrome to SARS-CoV-2 infection
New Down’s syndrome treatment improves cognition through epigenetic changes in the brain (29/09/2020)
CRG researchers describe some of the mechanisms underlying the potential beneficial effects of a new treatment
Dierssen lab discovers a therapy for Down syndrome (Lancet Neurology 07/06/2016)
New research published in Lancet Neurology has found using an inhibitor of DYRK1A , a serine/threonine kinase involved in brain plasticity, improves cognition in subjects with Down syndrome.
Summary
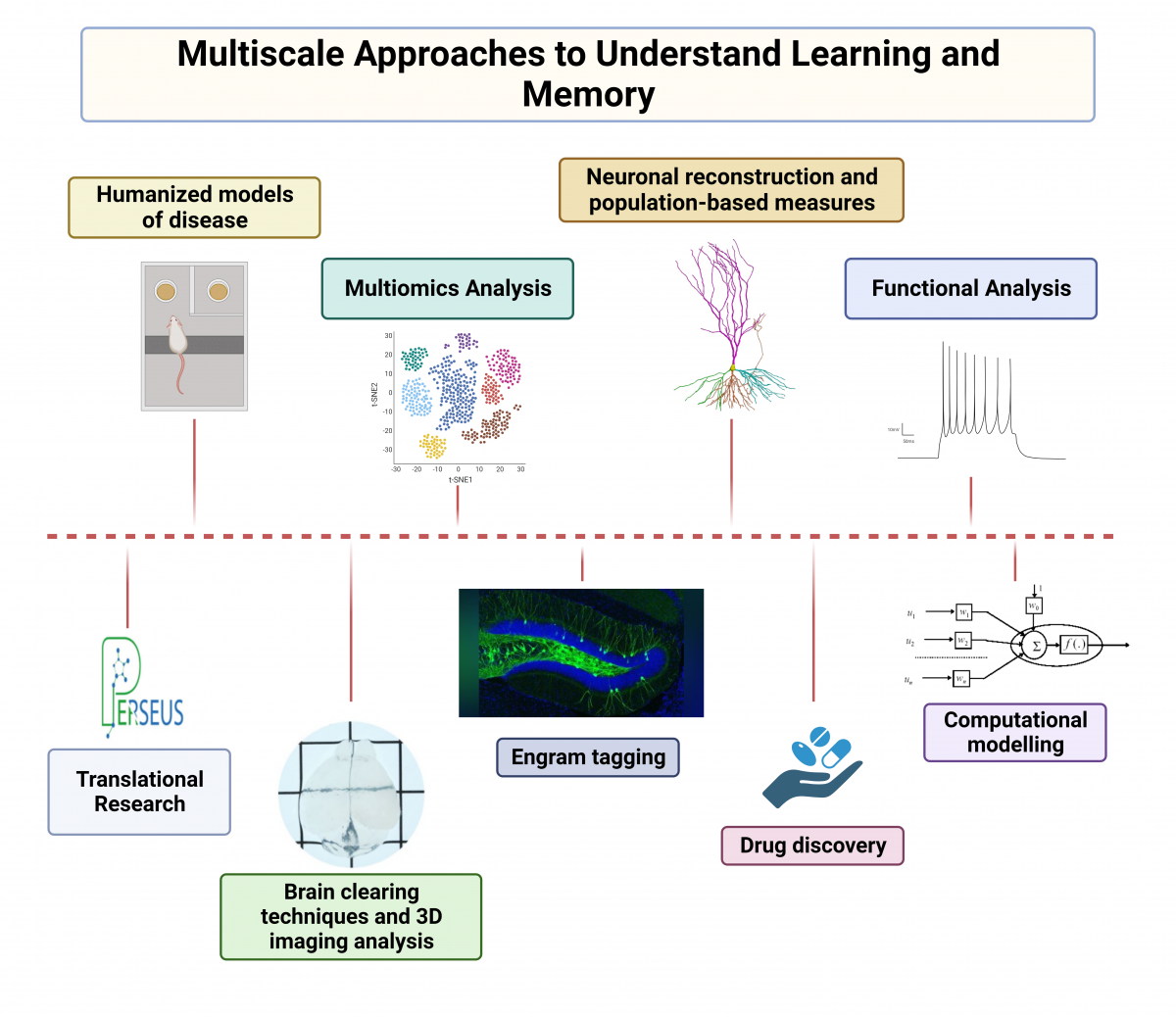
The Dierssen lab is centered on understanding the neurobiology of learning and memory and their perturbation in brain disorders. The central question we aim to address is how is memory coded in the brain.
To this aim we use a multi-disciplinary approach to understand how memories are encoded, stored, and retrieved in the brain, using a wide range of experimental techniques including engram tagging techniques, mouse transgenics, electrophysiology, calcium imaging, and behavioral analysis. We are also interested in the structural and functional properties of the neuronal networks underlying cognitive function, and in the molecular landscape sustaining those processes. In recent studies (PROMPT, EraPerMed), we also use multi-omics data (metabolomics, proteomics, transcriptomics, genomics) in an integrated precision medicine approach to major depression. The core objective is to develop a new algorithm for the prediction of treatment response which could be tested and validated in future clinical settings.
Other Activities
| TEDxUPF Tecnocampus Mataro (2023) |  |
| Dierssen lab co-organizes the Cajal Conferences 2022 |  |
Mara Dierssen elected president of the Spanish Brain Council
A window into the human brain. Mara Dierssen. CCCB
TEDxSantCugat - Mara Dierssen
| Brain Polyphony Concert |  |
TEDxSol - Mara Dierssen
The Institució CERCA launches a video that walks you through several types of conversation in a mock evaluation panel that hinder objectivity towards candidates
Funding acknowledgements

Title: Desarrollo de una terapia para el síndrome X frágil
Eligible total cost: 168.330 €
Period: 2020 –2024
Ref.: RTC2019-007230-1, RETOS COLABORACIÓN 2019
Funding agency: Ministerio De Ciencia e Innovación, Agencia Estatal de Investigación
Title: Obtaining antioxidant active ingredients from the carob pod and validation of its therapeutic potential (EURONUTRA)
Eligible total cost: 156.174,70 €
Period: 01/09/2020 to 31/12/2023
Ref.: RTC2019-007329-1, RETOS COLABORACIÓN 2019
Funding agency: Ministerio de Ciencia e Innovación, Agencia Estatal de Investigación
Title: Towards understanding and prediction of treatment resistance in major depression (Coordinator)
Eligible total cost: 399.985,75 €
Period: 01/05/2023 to 31/05/2025
Ref.: 202212-30-31-32
Funding agency: Marato TV3 Foundation
Title: Engramas de memoria en el síndrome de Down: alteraciones celulares y moleculares (PI)
Call: MCIU PE 2017-2020 I+D
Eligible total cost: 266.000 €
Period: 2020 –2023
Ref.: PID2019-110755RB-I00
Funding agency: Ministerio de Ciencia e Innovación, Agencia Estatal de Investigación
Title: Understanding interneuronopathy in Down syndrome to identify interneuron targeted therapy (INTO-DS)
Eligible total cost: 325.000 €
Period: 2023 to 2025
Ref.: PID2022-141900OB-I00
Funding agency: National Research Agency (Agencia Estatal de Investigación, AEI), from the Spanish Ministry of Science and Innovation.

“Psych-STRATA - A Stratified Treatment Algorithm in Psychiatry: A program on stratified pharmacogenomics in severe mental illness” has received funding from the European Union’s HORIZON-HLTH-2021 program under grant agreement No 101057454. 10.748.502.00 €. 2022-2027
Horizon 2020 (H2020-899986) “ICOD Innovative therapeutic approaches for cognitive impairment in Down syndrome” 6.105.089,50 €. 2021-2025,
EraPerMed (SLD044/20/000001) PROMPT “Toward PrecisiOn Medicine for the Prediction of Treatment response in major depressive disorder through stratification of combined clinical and -omics signatures 1.325.555 € 2021-2024
European Commission, Health (GO-DS21-848077) “Gene overdosage and comorbidities in Down Syndrome”, 5.965.967,75 €. 2020–2024




Title: "Ensayo clínico de Fase IIa de una terapia innovadora para pacientes de Síndrome de X Frágil"
Total budget: 271,725 €
Period: 01/05/2023 to 30/04/2026
Ref.: CPP2022-009659
Grant CPP2022-009659 funded by MCIN/AEI/ 10.13039/501100011033 and by “European Union NextGenerationEU/PRTR”.

The Research Project “Stratifying Cognitive Improvements to Reveal the Effect of Allelic Diversity in Trisomy 21” received the financial support of Jerome Lejeune Foundation.
Period: 10/01/2022 to 09/01/2024
Ref.: FJL2021_RCrans_Stratifying Cognitive Improvements to Reveal the Effect of Allelic Diversity in Trisomy 21
The Cellular and Systems Neurobiology Lab
There are two broad research programs in our lab. The first seeks to determine the organization and physiology of neural circuits in the brain involved with cognition and behavior and how they are perturbed in brain disorders. We integrate multi-faceted approaches from genetic engineering to quantitative neurobiology, integrating single-cell genomics, electrophysiology, imaging, neuroanatomy and quantitative behavioral analyses. Our second research program aims to link fundamental neuroscience to the understanding and treatment of neuropsychiatric and neurodegenerative disorders using multi-omic approached in human biofluids.
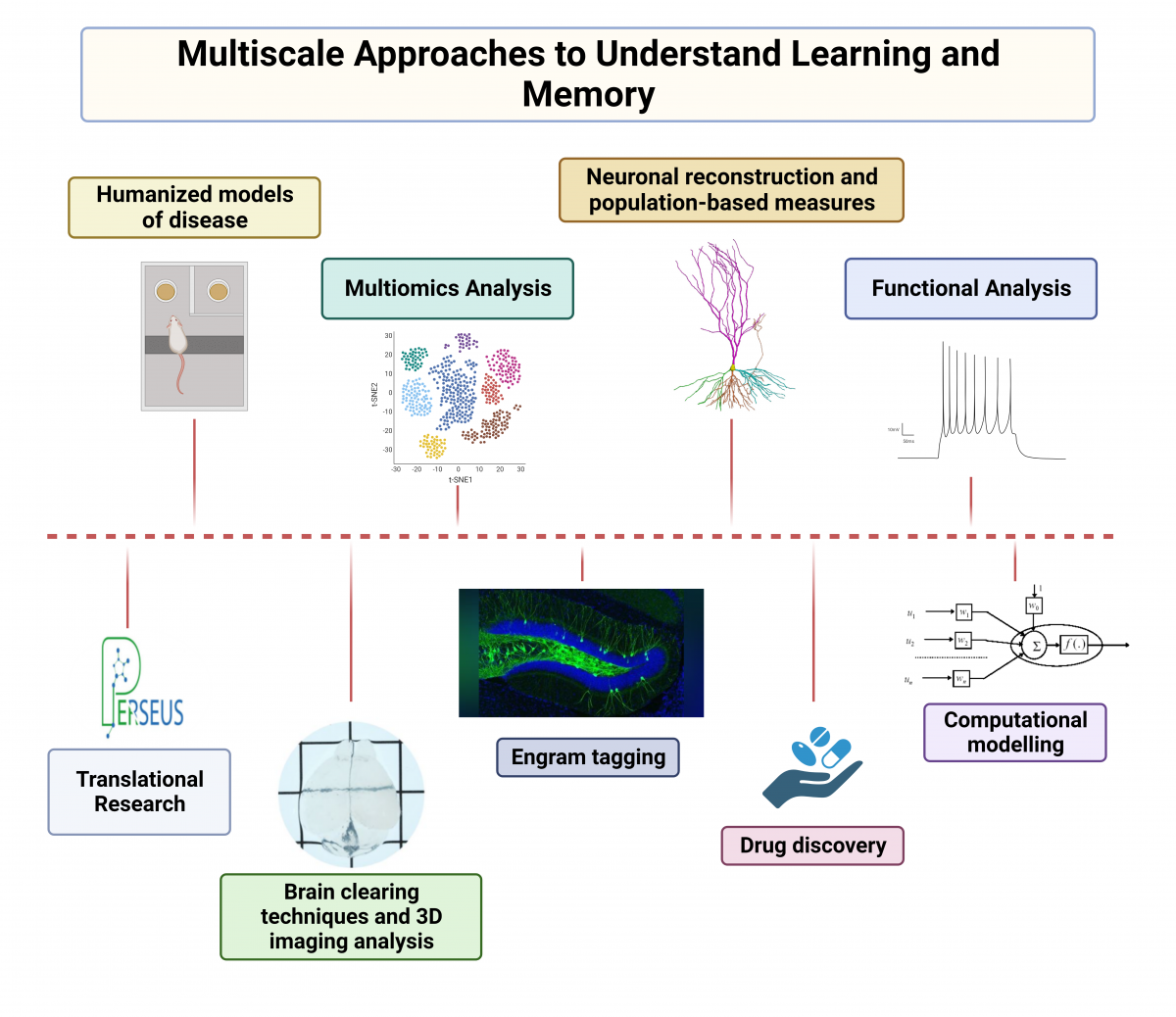
1. Cellular mechanisms underlying intellectual disability: engram pathology
Our aim is to understand how a stable memory trace is formed in the brain upon learning. We leverage on cutting-edge techniques like optogenetics and chemogenetics to achieve spatial and temporal resolution within specific sets of cells, so called engrams. Engrams are clusters of neurons that activate during the learning process and reactivate when memories are recalled. Our laboratory's research revealed impaired engram allocation in Down syndrome (DS) (Fernández-Blanco et al 2023). We have demonstrated that the overexpression of specific genes can result in an inherent interference with the establishment of stable memory traces (Ruiz-Mejias et al 2016; De Toma et al 2019; Ortega et al 2022). We also pioneered the discovery of new molecular players such as long non-coding RNAs or retrotransposons, a breakthrough that has revolutionized therapeutic approaches for cognitive disorders (Martinez de Lagran et al 2022; Sierra et al 2023). Distinct from other laboratories, we view astrocytes as an integral part of the synapse with roles in synapse function, and plasticity. We have discovered that activating astrocytes we rescue learning deficits in a mouse model of DS.
2. Translational Medicine and precision neuropsychiatry
We aim to identify potential candidate genes and regulatory elements that play pivotal roles in complex genetic conditions. Our genetic-dissection studies with DS mouse models pointed to the critical contribution of Dyrk1A overexpression to cognitive deficits in DS. This work led to the discovery of the first successful treatment for cognitive impairment in DS, which also increases functional brain connectivity (de la Torre et al 2017; 2020). This led to ten clinical trials in individuals with DS and Fragile X syndrome (Xicota et al, 2020; Cieuta-Walti et al, 2022). Furthermore, we have showed reduced functional connectivity on brain regions critical for learning and memory such as the prefrontal cortex and the hippocampus and, by harnessing the power of artificial intelligence, we are modelling the hippocampus to uncover mechanisms underlying its dysfunction in DS (Manubens-Gil et al 2023).
Our laboratory is embarking on an innovative trajectory, focusing on the fusion of pharmacogenomics and pharmaco-transcriptomics in neuropsychiatric disorders (Xicota et al, 2022; Pisanu et al 2022; Sathyanarayanan et al 2023). We focus on an urgent challenge in mental health: the emergence of drug resistance in a substantial number of patients. Through Horizon Europe and EraPerMed projects uniting genetic and genomic methodologies with AI-driven techniques, we aim to elucidate the biological mechanisms central to an individual's response to treatment and pave the way for therapies that are not only more effective but also more precisely aligned with the unique needs of each patient.





