Functional and pathological impact of microexons in endocrine pancreas
Functional and pathological impact of microexons in endocrine pancreas
Current lab members: Jonas Juan-Mateu, Marta Miret-Cuesta, Cristina Rodriguez, Luis P. Iñiguez, Jon Permanyer.
Former lab members: Simon Bajew, Amaya Lopez-Pascual.
Funding: ERC Consolidator Grant, La Caixa Health Research, EFSD/Lilly, CRG.
Diabetes is a metabolic disorder associated with severe clinical complications including cardiovascular disease. Its prevalence is increasing worldwide, representing a major health challenge. It results from dysfunction or loss of insulin-secreting pancreatic beta cells. While extensive research has uncovered the transcriptional networks that control differentiation and function of beta cells and showed that diabetes risk variants often disrupt cell type-specific enhancers, little is known about the roles of post-transcriptional regulation in beta cells. We found that a subset of neuronal microexons in also included in endocrine pancreas but not in most other tissues, including exocrine pancreas. Endocrine microexons, which we termed IsletMICs, impact a functionally coherent network of genes involved in vesicle transport and exocytosis, including genes associated with type 2 diabetes and insulin secretion. These microexons are regulated by the tissue-specific splicing factor SRRM3, whose depletion affects insulin release and response to diabetogenic stressors.
Interestingly, we observed that SRRM3 is consistently expressed at lower levels in pancreatic islets than in neurons, and that this is the key for the inclusion of only a subset of neural microexons in these cells: we found that IsletMICs are highly sensitive to the action of SRRM3, in contrast to neural-only microexons, which need much higher levels of SRRM3 for their inclusion. Therefore, microexons should no longer consider as a single program activated by a on/off switch (SRRM3/4), but they have at least two nested layers of inclusion, activated by different levels of the regulator (low: neural+pancreas, high: only neural).
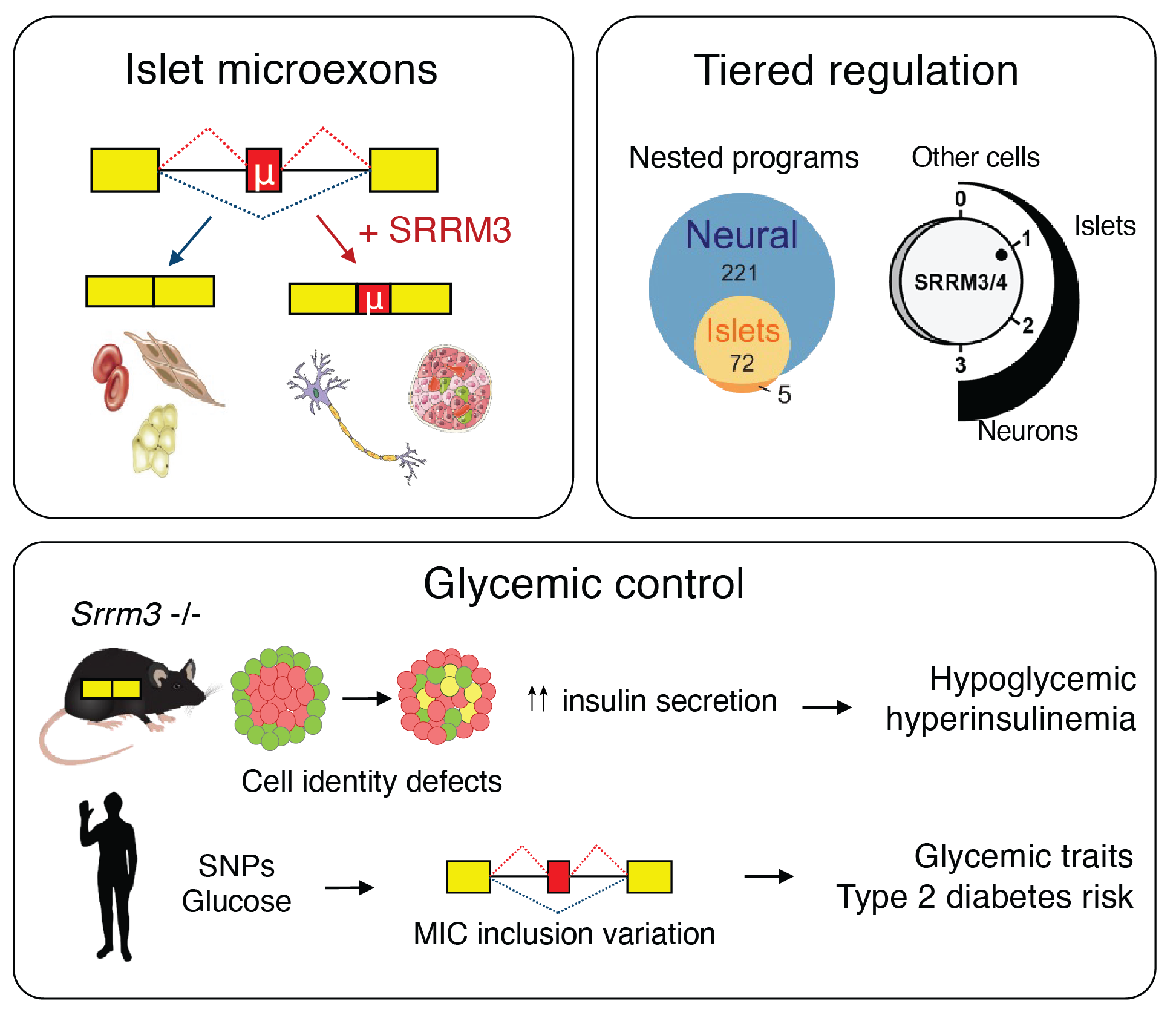
Graphical summary of IsletMICs
In this research line, we aim at investigating the mechanisms by which microexons impact insulin secretion and study their physiopathological role in glycemic control and diabetes. To address this, we: (i) study the bases of IsletMIC sensitivity to SRRM3, (ii) characterize the impact of microexons on insulin trafficking and exocytosis using beta cell lines; (iii) study the physiological role of the SRRM3-regulated microexon program on islet function and glucose metabolism using mouse and zebrafish models; (iv) assess their relevance for the development of endocrine pancreas in vertebrates; and (v) identify microexons associated with diabetes risk and assess their pathophysiological impact.
Altogether, this project will provide new insights into beta cell biology and generate valuable knowledge for the development of novel therapeutic targets and approaches in diabetes research.
Related publications:
- Juan-Mateu, J. †, Bajew, S., Miret-Cuesta, M., Íñiguez, L.P., López-Pascual, A., Bonnal, S., Atla, G., Bonàs-Guarch, S., Ferrer, J., Valcárcel, J.†, Irimia, M.† (2023). Pancreatic microexons regulate islet function and glucose homeostasis. bioRxiv. doi: 10.1101/2022.04.02.486809. Nat Metab, in press.
- Atla, G., Bonàs-Guarch, S., Cuenca-Ardura, M,, Beucher, A., Crouch, D.J.M., Garcia-Hurtado, J., Moran, I.; T2DSystems Consortium, Irimia, M., Prasad RB, Gloyn, A.L., Marselli, L., Suleiman, M., Berney, T., de Koning, E.J.P., Kerr-Conte, J., Pattou, F., Todd, J.A., Piemonti, L., Ferrer, J. (2022). Genetic regulation of RNA splicing in human pancreatic islets. Genome Biol. 23(1):196