 Mechanism of collagen secretion to address the issue of tissue fibrosis (collagenopathies)
Mechanism of collagen secretion to address the issue of tissue fibrosis (collagenopathies)
Dysregulated secretion and folding of ECM components are hallmarks of fibroses or the epidermolysis bullosa group of disorders. Initiation of collagen folding, targeting of misfolded collagens for degradation, and the selection of correctly assembled collagens for secretion, all take place in the endoplasmic reticulum but pathways that regulate these three processes are poorly understood. Our discovery of TANGO1 (Bard, Nature 2006; Saito, Cell 2009), a ubiquitously expressed, ER-exit-site-resident, transmembrane protein, has revealed the only known protein that stands at the crossroads of all these processes and could play a central role in directing ECM components down each path.
How does TANGO1 combine such diverse functions, and what does this multifunctional molecule tell us about membrane trafficking? TANGO1 and the TANGO1-related species TANGO1-like (TALI), are specifically required for the secretion of large cargoes such as collagens or chylomicrons that are too large to fit inside the originally described COPII vesicles of 60-90nm (Saito, Cell 2009; Saito, Mol Biol Cell 2011; Santos, J Cell Biol 2016; Malhotra, Ann Rev Cell Dev Biol 2015). Interactions between TANGO1 and cytoplasmic inner COPII coat protein Sec23A serve to gather, organize and link export machinery, with soluble cargo coordinately bound to TANGO1 in the ER lumen (Saito, Cell 2009; Ma, Proc Natl Acad Sci 2016; Ishikawa, Proc Natl Acad Sci 2016; Santos, J Cell Biol 2016).
Perhaps most important, is the interaction of TANGO1 with post-ER membranes, a phenomenon that has fundamentally altered our understanding of vesicle biogenesis and of the organisation of the early secretory pathway. Challenging a long-held dogma that vesicle biogenesis involves the consumption of membrane from one source organelle, we have seen that TANGO1 organises the site of vesicle formation – the ER exit site while dynamically physically linking two compartments, generating a carrier with membrane from both (Nogueira, eLife 2014; Santos, eLife 2015; Raote, J Cell Biol 2017).
Using every tool at our disposal, from in silico to structural and biochemical approaches to cutting-edge super resolution microscopy, working across model systems and organisms; we are working to arrive at a answer fundamental questions of the earliest events in the secretory pathway (Glick, J Cell Biol 2017).
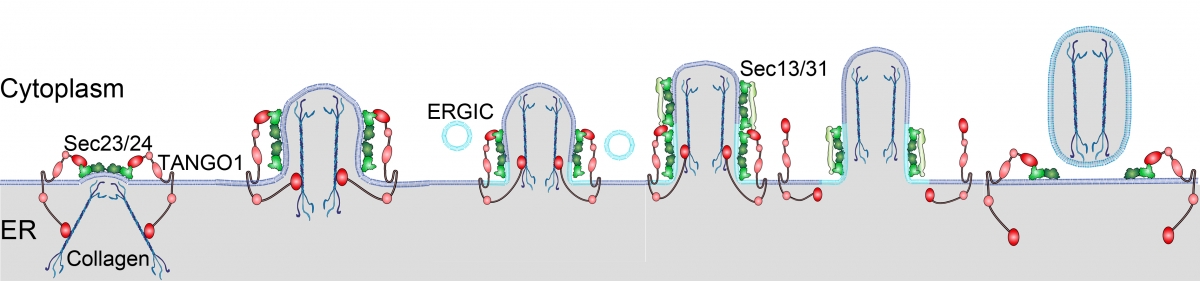
TANGO1 organisation in the export of collagen from the ER. TANGO1 interacts with collagen via Hsp47, bound to a lumenal SH3-like domain of TANGO1. The PRD is oriented inward, overlaying the inner COPII coat proteins. Stages I and II represent the initial growth by pushing of collagen against the lumenal face of the ER (II) and fusion of ERGIC-53–containing membranes. TANGO1 remains in a ring at the neck of the growing tubule, bound to Sec23 in the Sec23/Sec24 complex. Stage III corresponds to the growth of tubule that is large enough to accommodate collagens. Once this has been achieved, the separation of SH3-like domain from Hsp47/collagens in the lumen triggers the release of PRD from Sec23. This then follows the recruitment of Sec13/Sec31 to Sec23/ Sec24 at the neck of the collagen-filled tubule. Sec31-mediated activation of Sar1-GTP hydrolysis by Sec23 then leads to membrane scission to separate a collagen-filled container from the ER (Fission).
