 Advanced Light Microscopy Unit
Advanced Light Microscopy Unit
Advanced Light Microscopy Unit
Overview
The Advanced Light Microscopy Unit (ALMU) is a walk-in core unit on a mission to help, support and educate the research community from CRG and beyond to do quantitative bioimaging. We provide support on a range of technologies starting from experimental design, sample preparation, acquisition, bioimage analysis and visualization.
Our unit consists of microscope systems specifically designed for fluorescence microscopy ranging from widefield, and confocal to super-resolution systems and helps reveal structures from macro to the nanoscale. Together with our next-door neighbors, the Mesoscopic Imaging Facility (MIF) of the EMBL Barcelona, we cover the full spectrum of light microscopy and we collaborate to assist the scientific community in advancing their research. Click on the services tab to learn more about our support and how to gain access to our instruments.
You can learn more about the unit by taking the virtual tour HERE.
Latest Updates
| ALMU welcomes a new staff member Giovanni De Angelis (15/04/2026) Join us in welcoming Giovanni De Angelis to the team! Giovanni, a former Ph.D. student in the Biophysical Imaging group of Prof. Eggeling in Jena, Germany, joins us as an Imaging Scientist. After graduating in Physics Engineering from the Politecnico of Milan, he moved to Jena for his Ph.D., where he gained experience in implementing custom-made optical setups, fluorescence and scattering-based optical microscopy techniques with a focus in single-molecule-sensitive and super-resolution methods. We're excited to work with Giovanni and look forward to the contributions he will bring to ALMU. |
|
 |
Euro-BioImaging Kick-off meeting (20/02/2026) This event marks an important milestone for the consolidation of Euro-BioImaging Spain as part of the European research infrastructure, bringing together the national imaging community, node representatives, international experts, and institutional stakeholders. For more information and registration follow the link here. |
| ALMU welcomes new members (02/02/2026) Join us in welcoming Dr. Laura Bianchi, Rita Németh and Guillem Romero Camó to the team! Laura, a former CRG Ph.D. student and ALMU star user, joins us for a four-month period to help bridge a staffing gap. Rita joins for one year period, having graduated from Eötvös Loránd University and experience with imaging and image analysis, will help elevate ALMU’s workflows and QC measurements. Guillem joins with previous experience as an imaging scientist at the Institute for Bioengineering of Catalonia (IBEC). We're excited to work with all new members and look forward to the contributions they will bring to ALMU. |
|
 |
We are hiring! We are looking for a new team member with a background in physics or a related field. Experienced in building bespoke and adapting microscope systems and knowledge of python is required. Other experiences with optical microscopy, e.g. SMLM, confocal and their applications would be a +. For more information click here. |
| New Spinning disk confocal microscope with FRAP & super-resolution capabilities at ALMU We are delighted to offer the Evident/Olypmus IXplore SpinSR spinning disk confocal microscope as part of our portfolio. We've named it "SpinSR Live" to reflect its purpose. The SpinSR Live is equipped with 6 laser lines (405, 445, 488, 514, 561 and 640 nm), a 30x/1.05 NA and a 60x/1.3 NA silicon objective for live-cell imaging, 2 Hamamatsu Fusion BT CMOS cameras, and the SoRa system. Additionally, it includes a FRAP unit with 405, 488, and 640 nm lasers, and a full environmental chamber for temperature, CO2 and humidity control. For more information, training on the system, or testing, please send us an Agnedo request or contact ALMU (almu@core.crg.eu). |
|
 |
True Image Deconvolution, Restoration, and Analysis Workshop (13/03 & 03/04/2025) Join us for the upcoming True Image Deconvolution, Restoration, and Analysis workshop run by the Scientific Volume Imaging (SVI). This course will focus on obtaining high-quality microscopy images for reliable analysis results. Topics covered will include diffraction, acquisition pitfalls, spherical aberration, photon noise, Point Spread Function, Nyquist-Shannon Sampling Rate, Image Quality Control, crosstalk, (colocalization) analysis, and deconvolution. For more information follow the link here. Deadline for registration March 10, 2025. |
 |
Lattice SIM 3 Launch Tour starting at ALMU (22-30/04/2024) ALMU is collaborating with Carl ZEISS Iberia to host the new Lattice SIM 3 system first landing in Spain. The system is designed to meet the imaging requirements of multicellular organisms and tissue sections. To register follow the link here. April 29: Virtual seminar covering the Lattice SIM 3 system capabilities and highlighting the technology. Followed by a virtual live demo of the system. |
 |
Quantitative Light Microscopy Course - QLM2024 (22-26/04/2024) ALMU together with the CRG training team is organizing the second edition of the Quantitative Light Microscopy Course, QLM2024. This unique course covers the entire spectrum of bioimaging, starting from the fundamental basis of optical microscopy, instrumentation, specimen preparation, molecular markers, right up to the most recent and revolutionary development in bioimaging and bioimage data analysis, including artificial intelligence in microscopy along with hands-on practical sessions. For more information follow the link here. |
|
ALMU welcomes a new staff member Sanni Erämies (15/01/2024) Join us in welcoming Sanni Erämies to the team! Sanni steps in during the one-year leave of Arrate Mallabiabarrena Ormaechea. With Sanni's previous experience as an imaging scientist at the Tampere Imaging Facility, Uni. of Tampere, we're excited to work with her and for the contributions she'll bring to ALMU. |
|
 |
The Kingdome of Spain has joined Euro-Bioimaging (14/12/2023) ALMU became part of two Spanish nodes for Euro-BioImaging: SLN@BCN - Barcelona Super Resolution Light Nanoscopy - Super Resolution Node, and Barcelona Live and Intravital - Advanced Light Microscopy Node (BLivIN). For more information follow the link here. |
 |
OMERO Workshop (7-8/11/2024) ALMU together with OME, the University of Dundee, and CRG's SIT & CRG training are organizing the OMERO workshop. This course will give a solid ground for CRG researchers and all researchers who would like to use the open-source image data management software to enable access, analysis, visualization, sharing, and publishing of microscopy data. This course is also an important milestone for CRG to start using CRG's OMERO server for image data management. For more information follow the link here. |
 |
Spotlight on Jeremy Vincencio, Shoma Nakagawa & ALMU in Evident's innovative laser scanning confocal system brochure We are proud to see the fruit of our successful collaboration with Evident and ALMU users: Jeremy Vincencio's (Stroustrup Lab) and Shoma Nakagawa (Cosma Lab) in the brochure of Evident's new innovative laser scanning confocal system FLUOVIEW FV4000. Tests on the prototype were carried out at ALMU. For more information follow the link here. |
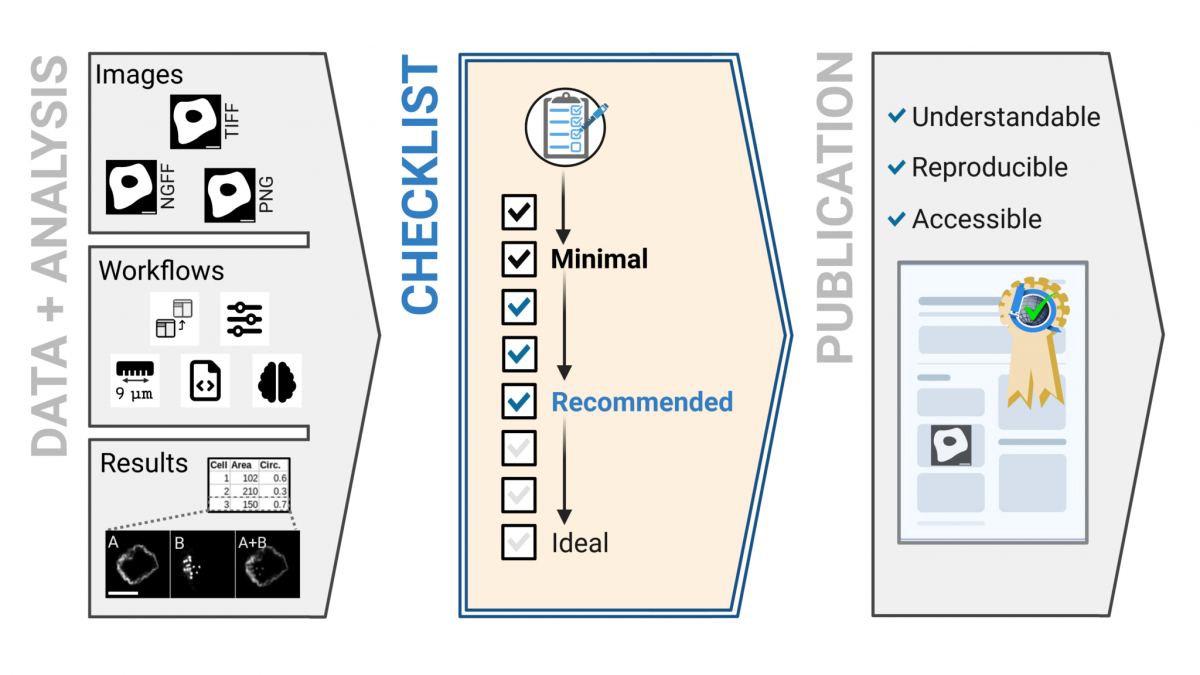 |
ALMU collaborates with a global initiative to promote reproducible microscopy data publishing (21/09/2023) |
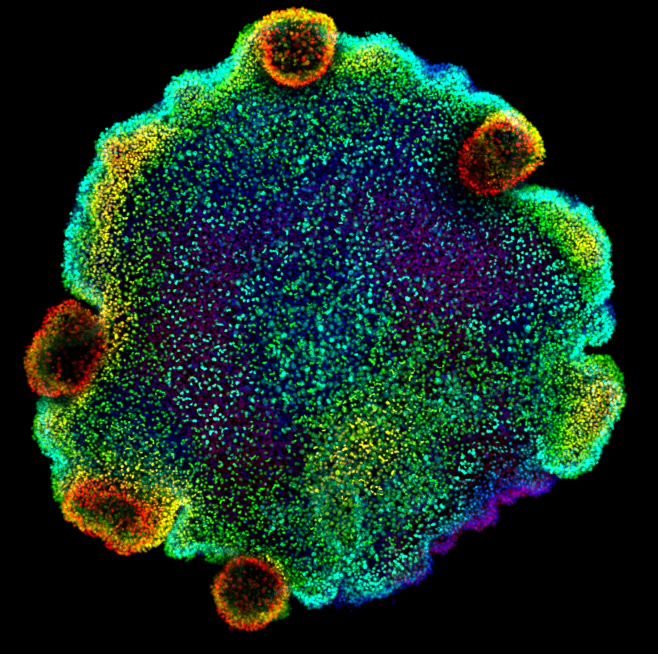 |
ALMU collaborates to reveal the ancient origins of neurons (19/09/2023) |
ALMU collaborates to test the new function Dynamic Profiler from Carl Zeiss
We are proud to see the successful launch of the new function Dynamic Profiler which enables the measurement of molecular dynamics with spatial information. Tests were carried out at ALMU, with ALMU and ALMU users. For more information follow the link here.
Elyra 7 with Lattice SIM2 workshop with Carl Zeiss (27/03/2023)
ALMU hosted the Elyra 7 workshop that presents the latest advances allowing gentle imaging of cellular structure in the nanoscale, using SIM, PALM/STORM technologies at high speed with the ability to image deeper into the samples.
February 27: Virtual seminar covering the ELyra 7 system capabilities and highlighting the super-resolution technology. Followed by a virtual live demo of the system.
February 27 - March 3: Hands-on demo with researcher's own samples.
 |
Quantitative Light Microscopy Course (20-24/02/2023)
|
IXplore SpinSR workshop with Evident/Olympus (04/05/2022)
ALMU hosted the IXplore SpinSR workshop that presented the latest advances in multi-modal confocal spinning disk imaging of live cells and tissues in super-resolution mode.
May 4: Virtual seminar covering the IXplore SpinSR system capabilities and highlighting the super-resolution technology. Followed by a virtual live demo of the system.
May 5 - May 18: Hands-on demo with researcher’s own samples.
CRG at the Dubai Expo Spain Pavilion (03/2022)
Nadia Halidi has joined forces with Neoalgae, an Austrian biotechnology company, and The Others TV, an animation studio based in Barcelona to showcase the potential of microalgae as a sustainable future solution. The movies were acquired with a high-speed camera using darkfield microscopy with components provided specifically for this project by Carl Zeiss Iberia S.L. The production was showcased in the Spain Pavilion at the Expo 2020 Dubai from October 2021 to March 2022.
Nadia Halidi CV
February 2021 Head of the Advanced Light Microscopy Unit, CRG, Barcelona, Spain.
2018 - 2021 Assistant manager, then manager of Micron Oxford Advanced Bioimaging Unit, University of Oxford, Oxford, UK.
2017 - 2018 Postdoctoral researcher and imaging facility manager at the Botnar Research Center, University of Oxford, Oxford, UK.
2011 - 2015 Postdoctoral fellow at the Dept. of Cell Biology, Harvard Medical School, Boston, MA, USA.
2007 - 2011 PhD in Biophysics, École Polytechnique Fédérale de Lausanne (EPFL), Lausanne, Switzerland.
2005 Teaching assistant at the Kalamazoo College, Kalamazoo, MI, USA.
2002 - 2006 Research and teaching assistant at the Dept. of Biophysics, University of Pécs, Pécs, Hungary.
Services
- Consultation and assistance starting from experimental design, sample preparation, imaging, and data analysis
- Technology training on the operation of the unit’s microscope systems
- Support for experiments requiring advanced imaging methods
- User access to state-of-the-art microscopy systems
- User access to analysis workstations
- Image data analysis services
How to access / Instrument Booking
The Advanced Light Microscopy Unit (ALMU) is open to the research community from within CRG, the PRBB, and beyond. ALMU houses a range of state-of-the-art microscopes, from widefield, confocal to super-resolution systems. We offer training, instrumentation, and support for experimental design, sample preparation, data analysis, and visualization to the highest standards.
To request access and training, please read our Access Policy. Due to the high demand and to ensure that the training goes smoothly, please contact us 1-2 weeks in advance.
For existing users, all our resources can be booked through our booking system AGENDO: https://crg.agendoscience.com/
Service Prices
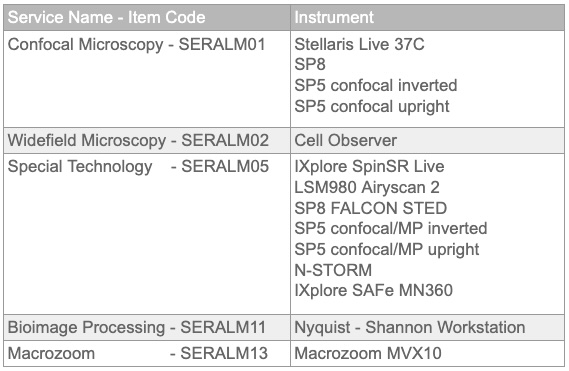
Equipment
The equipment offer is available to trained users and covers all current imaging applications with high-end imaging systems.
Super-resolution microscopy
- Leica confocal STED 3X FALCON system with white light laser (WLL) and three depletion wavelengths (WLL: 470 - 670 nm, 592nm CW, 660nm CW, 775nm pulsed)
- Nikon N-STORM (405nm, 488nm, 561nm, 647 nm lasers)
- Zeiss LSM 980 with airyscan 2 (405, 488, 561, 639 nm lasers)
- Evident/Olympus SpinSR spinning disk confocal system with super-resolution and FRAP (405, 445, 488, 514, 561, 640 nm lasers, for FRAP: 405, 488 and 640 nm lasers) for live-cell imaging.
Multiphoton microscopy
- Leica inverted confocal/multiphoton system (405, Argon ion, 561, 633nm and Mai Tai DeepSee)
- Leica upright confocal/multiphoton system (405, Argon ion, 543, 633nm and Mai Tai DeepSee)
Confocal Microscopy
- Evident/Olympus SpinSR spinning disk confocal system with super-resolution and FRAP (405, 445, 488, 514, 561, 640 nm lasers, for FRAP: 405, 488 and 640 nm lasers) for live-cell imaging.
- Leica STELLAIRS confocal system with white light laser ( WLL: 485 - 685, also 405, 488, 561 nm lasers)
- Leica inverted confocal system (405, 458,476,488,496,514,561 and 633 nm lasers)
Widefield Microscopy
- Zeiss Cell Observer fluorescence microscope for live-cell imaging
- Olympus MVX10 Macro Zoom fluorescence microscope
Computer resources & software packages
- Bioimage analysis workstation for processing and analysis (Processor: Intel(R) Xeon(R) W-2255 CPU @ 3.70GHz, RAM: 128 GB and GPU: NVIDIA RTX A6000).
- Open-source software available for users: ImageJ/Fiji, CellProfiler, Advanced Cell Classifier, ilastik, cellpose, napari, R studio.
- Commercial software available for users: SVI Huygens Professional, and Imaris 3/4D Bioimage Visualization Software (Oxford Instruments).
- OMERO














