The CRG is an international biomedical research institute of excellence...
More information
 News
News
12
Mayo
CRG researchers call for protein-design systems to be more transparent, trustworthy and secure
12
Mar
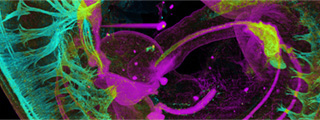
A CRG study reveals a new way of measuring splicing directly
06
Mar
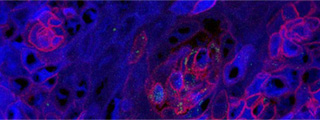
First evidence that the nucleus is crowded with metabolic enzymes
 Events
Events
28
Mayo
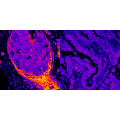
The human body consists of trillions of cells, but a mutation in one of them can cause cancer.
This example demonstrates that biology is not a process driven by averages and highlights the importance of single cell-level studies. The last years have...
22
Sep
La Alianza de Centros de Excelencia Severo Ochoa y Unidades de Excelencia María de Maeztu (SOMMa) junto al Centro de Regulación Genómica (CRG) organizan el evento SOMMa Connect que tendrá lugar el 22 y 23 de septiembre de 2026 en el auditorio del...
27
Oct
The 2026 CRG Symposium will explore how cells are organized, evolve, and function across scales, integrating perspectives from biophysics, cell biology, evolution, and advanced imaging. By bringing together leading researchers from the cell biology...